UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of report (Date of earliest event reported): March 12, 2020
Precision BioSciences, Inc.
(Exact name of registrant as specified in its charter)
|
Delaware |
|
001-38841 |
|
20-4206017 |
|
(State or other jurisdiction of incorporation) |
|
(Commission File Number) |
|
(IRS Employer Identification No.) |
302 East Pettigrew St., Suite A-100, Durham, North Carolina 27701
(Address of principal executive offices) (Zip Code)
(919) 314-5512
(Registrant’s telephone number, including area code)
N/A
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
|
|
☐ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
|
|
☐ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
|
|
☐ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
|
|
☐ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
|
Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered |
|
Common stock, par value $0.000005 per share |
DTIL |
The Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01. Regulation FD Disclosure.
As previously announced, Precision BioSciences, Inc. (the “Company”) will be presenting at the Barclays Global Healthcare Conference in Miami, Florida on March 12, 2020. A copy of the accompanying presentation materials that the Company will discussing in meetings with investors and analysis is furnished as Exhibit 99.1 hereto and is incorporated herein by reference. These presentation materials are also available on the Investor Relations page of the Company’s website at https://investor.precisionbiosciences.com.
The information in Item 7.01 of this Current Report on Form 8-K (including Exhibit 99.1) shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that Section, nor shall it be deemed to be incorporated by reference into any filing of the Company under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly set forth by specific reference in such filing.
Item 9.01.Financial Statements and Exhibits.
|
|
(d) |
Exhibits |
|
|
|
|
|
Exhibit |
|
Description |
|
|
|
|
|
99.1 |
|
Precision Biosciences, Inc. Presentation as of March 12, 2020 |
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
|
|
|
|
|
|
|
|
|
PRECISION BIOSCIENCES, INC. |
||
|
|
|
|
|
|||
|
Date: March 12, 2020 |
|
|
|
By: |
|
/s/ Abid Ansari |
|
|
|
|
|
|
|
Abid Ansari |
|
|
|
|
|
|
|
Chief Financial Officer |

March 2020 Exhibit 99.1

This presentation (together with any other statements or information that we may make in connection herewith) may contain forward-looking statements. All statements other than statements of present and historical facts contained in this prospectus, including without limitation, statements regarding our future results of operations and financial position, business strategy and approach, including related results, prospective products, planned preclinical or greenhouse studies and clinical or field trials, including expected release of data and dosage exploration, capabilities, including expected production levels and manufacturing timeframes, of our manufacturing facility, management’s expectations regarding pipelines and milestones for product candidates and our food editing platform, and timing and likelihood of success, as well as plans and objectives of management for future operations, may be forward-looking statements. Without limiting the foregoing, the words “aim”, “anticipate,” “believe,” “could,” “expect,” “should,” “plan,” “intend,” “estimate,” “target,” “may,” “will,” “would,” “potential,” the negative thereof and similar words and expressions are intended to identify forward-looking statements. These forward-looking statements reflect various assumptions of Precision’s management that may or may not prove to be correct. No forward-looking statement is a guarantee of future results, performance, or achievements, and one should avoid placing undue reliance on such statements. Forward-looking statements are based on our management’s beliefs and assumptions and on information currently available to us. Such statements are subject to a number of known and unknown risks, uncertainties and assumptions, and actual results may differ materially from those expressed or implied in the forward-looking statements due to various factors, including, but not limited to: our ability to become profitable; our ability to procure sufficient funding and requirements under our current debt instruments; our limited operating history; the success of our programs and product candidates in which we expend our resources; our dependence on our ARCUS technology; the initiation, cost, timing, progress and results of research and development activities, preclinical or greenhouse studies and clinical or field trials; our or our collaborators’ ability to identify, develop and commercialize product candidates; our or our collaborators’ ability to advance product candidates into, and successfully complete, clinical or field trials; our or our collaborators’ ability to obtain and maintain regulatory approval of our product candidates, and any related restrictions, limitations and/or warnings in the label of an approved product candidate; the laws and regulatory landscape applicable to our and our collaborators’ development of product candidates; our ability to achieve our anticipated operating efficiencies at our manufacturing facility; delays or difficulties in enrolling patients in clinical trials; our ability to obtain and maintain intellectual property protection for our technology and any of our product candidates; potential litigation relating to infringement or misappropriate of intellectual property rights; if our product candidates do not work as intended or cause undesirable side effects the potential for off-target editing or other adverse events, undesirable side effects or unexpected characteristics associated with any of our product candidates; risks associated with applicable healthcare, data privacy and security regulations and our compliance therewith; the rate and degree of market acceptance of any of our product candidates; the success of our existing collaboration agreements; our ability to enter into new collaboration arrangements; public perception about genome editing technology and its applications; competition in the genome editing, biopharmaceutical, biotechnology and agricultural biotechnology fields; potential manufacturing problems associated with any of our product candidates; pending and potential liability lawsuits and penalties related to our technology and our product candidates; our reliance on and current and future relationships with third parties; our ability to effectively manage the growth of our operations; our ability to attract, retain, and motivate key scientific and management personnel; effects of natural or manmade disasters; insurance expenses and exposure to uninsured liabilities; market and economic conditions; dilution and fluctuations in our stock price; and other important factors discussed under the caption “Risk Factors” in our Annual Report on Form 10-K for the annual period ended December 31, 2019, as such factors may be updated from time to time in our other filings with the SEC, which are accessible on the SEC’s website at www.sec.gov. All forward-looking statements speak only as of the date of this presentation, and except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. This presentation may also contain estimates, projections, and/or other information regarding our industry, our business and the markets for certain of our product candidates, including data regarding the estimated size of those markets, and the incidence and prevalence of certain medical conditions. Unless otherwise expressly stated, we obtained this industry, business, market and other data from reports, research surveys, clinical trials, studies and similar data prepared by market research firms and other third parties, from industry, medical and general publications, and from government data and similar sources. Information that is based on estimates, forecasts, projections, market research, or similar methodologies is inherently subject to uncertainties and actual events or circumstances may differ materially from events and circumstances reflected in this information.

Delivering on the Promise of Genome Editing ARCUS genome editing platform built for translation with full freedom to operate In vivo gene correction platform seeking to cure genetic and infectious diseases Wholly integrated food editing platform focused on human wellness and food security Scaled in-house cGMP manufacturing Clinical stage allogeneic CAR T platform with validating initial safety and response data World class team that includes the pioneers in genome editing Strong balance sheet: $181m cash* funds into 2H 2021 * As of FYE 2019

ARCUS: Precision’s Proprietary Gene Editing Platform Precision bioengineering I-CreI ARCUS Key Advantages Safety: minimizes off-target editing; natural “off switch” Ease of delivery: small size permits both LNP and AAV delivery Control of edits: efficient knock in or knock out Proprietary: more than 50 issued US and foreign patents Nature’s editing system, evolved for highly precise, versatile gene editing I-CreI engineered to target desired genetic sites

Efficient, Scaled In-house Manufacturing Already Established Operational July 2019 17,300 sq. ft. facility in Durham, NC Currently producing clinical trial material for BCMA CAR T program Fully cGMP compliant Ability to produce ARCUS-based CAR T and in vivo therapies MCAT: Manufacturing Center for Advanced Therapeutics

Overcoming Cancer Off-the-Shelf CAR T

The Allogeneic CAR T Opportunity Autologous CAR T Allogeneic CAR T High rates of efficacy in some cancers Can be effective where other options have failed Early evidence of encouraging clinical efficacy High cost and challenging logistics leading to limited patient access Patient-to-patient product variability Safety considerations (CRS / neurotoxicity) Potentially widely available and lower cost Standardized product profile Potential for dose optimization like “traditional” drugs Potential for improved safety profile Opportunity to benefit many more patients Patient draw Single treatment time Ready when needed

True off-the-shelf safety profile Supports ease of use / physician adoption Minimize CRS / neurotoxicity Efficiently deliver consistent, high quality cell product Reach all eligible patients Control costs Healthy cell product High percentage naïve and central memory T cell phenotypes Ability to employ more tolerable conditioning regimens Demonstrated efficacy Ability to optimize dosing Four Key Requirements for Allogeneic CAR T Success Optimal T Cell Phenotype 2 Improved Safety 3 Clinical Activity 4 Scaled Manufacturing 1

Single-step ARCUS editing minimizes off-targeting CAR directly inserted into TCR locus helps preserve phenotype Key Features of Precision’s Allogeneic CAR T Platform Optimized T cell from healthy donor High yield, consistent drug product targeted Short, 10-day manufacturing CAR expressed with proprietary N6 co-stimulatory domain TCR knocked out CAR knocked in TCR

>50% naïve and central memory T cells Zero GvHD, severe CRS or neurotox; zero infections* Ability to manufacture consistent product at scale Encouraging early activity at low dose levels* Platform Is Delivering Against All Four Key Requirements 67% ORR in NHL 2 CRs achieved 28+ days 1 NHL / 1 ALL 180 day PFS observed at low dose Optimal T Cell Phenotype 2 Safety Profile 3 Clinical Activity 4 Scaled Manufacturing 1 % CAR+ Cells NHL (n=6) ALL (n=3) GvHD DL1 0% 0% DL2 0% 0% CRS/ICANS ≥ Grade 3 DL1 0% 0% DL2 0% 0% Infections DL1 0% 0% DL2 0% 0% *Clinical data from PBCAR0191 DL1 & DL2 interim update presented in December 2019; n=6 NHL patients and n=3 ALL patients Final Yield CD19 Drug Candidate (64M CAR T cells/vial) CD3- >99% Batch Vial Count 1 130 2 114 3 100 TN/SCM TCM

Precision’s Off-the-Shelf CAR T Immunotherapy Pipeline Product Candidates Program Area Discovery Pre-clinical Clinical Rights PBCAR0191 (CD19) PBCAR20A (CD20) PBCAR269A (BCMA) NHL and ALL – Phase 1 Data Expected 2020 NHL, CLL, SLL – Phase 1 Dosing Expected Q1 2020 MM - IND cleared – Phase 1 Dosing Expected 2020 Three allogeneic CAR T programs expected to be in clinical trials in 2020

PBCAR0191 Phase 1/2a: NHL and B-ALL -14 -7 -5 -4 -3 0 28 60 90 180 360 Screening Follow-Up Day LTFU Study Treatment Period PBCAR0191 Infusion x1 Enrollment Safety & Response Assessment End of Study Lymphodepletion Safety Assessment Population Adult patients with R/R B-NHL or R/R B-ALL Objectives Primary: safety and tolerability Secondary: anti-tumor activity Exploratory: expansion, trafficking, and persistence Dose Escalation Dose range: 3.0 x 105 cells/kg - 9.0 x 106 cells/kg Lymphodepletion regimen Fludarabine 30 mg/m2/day + Cyclophosphamide 500 mg/m2/day

Adverse Events Compare Favorably to Autologous CAR T System Organ Class Preferred Term, n(%) NHL (n=6) B-ALL (n=3) CRS (Cytokine Release Syndrome) – Grade 1 or Grade 2 2 (33%) 1 (33%) ICANS (Immune Effector Cell Neurotoxicity) – Grade 1 or Grade 2 0 (0%) 1 (33%) CRS Grade 3 or higher 0 (0%) 0 (0%) ICANS Grade 3 or higher 0 (0%) 0 (0%) GvHD (Graft versus Host Disease) 0 (0%) 0 (0%) Infection 0 (0%) 0 (0%)

Initial Data Support Dose Dependent Clinical Activity NHL Cohort Data Best response day 28+ # patients DL1 (n=3) DL2 (n=3) CAR T cell expansion # patients with positive expansion (maximum days positive) 2/3 pts (Day 3**) DL1 (n=3) DL2 (n=3) qPCR Flow qPCR Flow 2/3 pts (Day 21†) 3/3 pts (Day 60†) 0/3 pts 67% ORR Note: For presentational purposes, ALL cohort data not illustrated. In ALL cohort (n=3, treated at DL2), best response observed at day 28+ of 1 patient with complete response and 2 patients with progressive disease * Expansion formally reported as below lower limit of quantification by qPCR for this patient, but was detectable at day 7 ** Ranged from day 1 to day 3 † Ranged from day 10 to day 21 by qPCR; day 1 to day 60 by flow 1/3 pts* 2 Partial responses 1 Partial response 1 Complete response
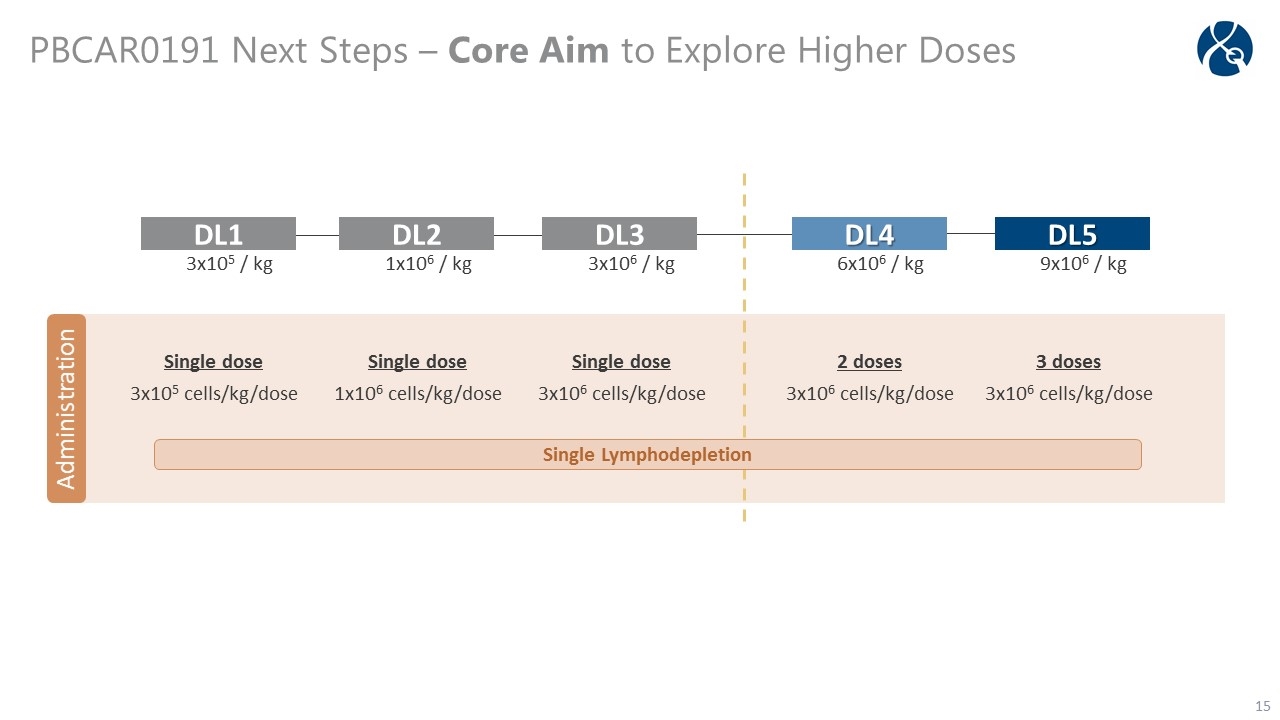
PBCAR0191 Next Steps – Core Aim to Explore Higher Doses Administration DL5 9x106 / kg DL4 6x106 / kg DL3 3x106 / kg DL2 1x106 / kg DL1 3x105 / kg Single dose 3x105 cells/kg/dose Single dose 1x106 cells/kg/dose Single dose 3x106 cells/kg/dose 2 doses 3x106 cells/kg/dose 3 doses 3x106 cells/kg/dose Single Lymphodepletion

PBCAR20A and PBCAR269A Allogeneic CAR T Programs Targeting CD20 PBCAR20A Targeting BCMA PBCAR269A Population Dose Escalation Status Adult patients with R/R NHL (including MCL) or R/R CLL or SLL DL1 = 3.0 × 105 cells/kg DL2 = 1.0 × 106 cells/kg DL3 = 3.0 × 106 cells/kg Phase 1/2a expected to begin Q1 2020 ODD granted for MCL Adult patients with R/R multiple myeloma DL1 = 6.0 × 105 cells/kg DL2 = 2.0 × 106 cells/kg DL3 = 6.0 × 106 cells/kg IND cleared January 2020 Phase 1/2a expected to begin in 2020 Screening Follow-Up Day LTFU Study Treatment Period CAR T Infusion x1 Enrollment Safety & Response Assessment End of Study Safety Assessment Lymphodepletion Fludarabine 30 mg/m2/day + Cyclophosphamide 500 mg/m2/day -14 -7 -5 -4 -3 0 28 60 90 180 360 Objectives Primary: safety and tolerability Secondary: clinical (anti-tumor) activity Exploratory: expansion, trafficking, and persistence FDA approval to begin at DL2 - based on PBCAR0191 safety profile

Off-the-Shelf CAR T: The Best of Both Worlds Off-The-Shelf Biologics First Line Safety profile Accessibility CAR T Last Line Efficacy profile Future portfolio in solid tumors Liquid tumors ü Safety profile Accessibility Efficacy profile PBCAR

Curing Genetic Disease In Vivo Gene Correction

Safety 1 Editing Drives Therapeutic Effect 4 Ease of Delivery 2 Efficient, Versatile Editing 3 Four Key Requirements for Successful In Vivo Gene Correction Knock in and/or repair of gene defects substantially increases utility of platform Specific and high efficiency Minimize off-target gene editing Critical for viability of in vivo editing Delivery vectors have limited capacity Smaller payloads expand range of delivery options Increase potential number of addressable organs / diseases High efficiency editing corresponds to therapeutic effect Large animal data critical Editing platform must be designed for clinical translation

Safety 1 ARCUS Platform Delivers Against All Four Key Requirements Full freedom-to-operate for ARCUS platform * as assessed by oligo capture technique PCSK9 data reported in part in Wang et al, Nature Biotechnology, 2018 Gen 3 ARCUS achieved undetectable levels of off target editing in NHPs Gen 3 n=3 NHPs 0 # off-target sites* PCSK9 ARCUS - NHP off-targeting assessment Gen 1 n=4 NHPs ARCUS PCSK9 knock out delivered durable therapeutic effect in NHPs Editing Drives Therapeutic Effect 4 ~90% reduction in serum PCSK9 levels in NHPs ARCUS easily “fits” in established vehicles like AAV Ease of Delivery 2 ARCUS achieved high-efficiency PCSK9 gene knock out Efficient, Versatile Editing 3 Frequency of ARCUS PCSK9 liver editing events in NHP Control Knock out Day 128

Product Candidate Program Area Discovery Pre-clinical Clinical Rights HBV HAO1 Transthyretin ApoC3 PCSK9 Familial amyloid polyneuropathy Chronic Hepatitis B – IND 2021 Lipoprotein lipase deficiency Familial hypercholesterolemia In Vivo Gene Correction Pipeline PH1 selected as lead wholly-owned in vivo program Primary hyperoxaluria type 1 (PH1)

Overview of Primary Hyperoxaluria Type 1 (PH1) Prevalence of 1-3/1,000,000 Affects adults and young children ~40% patients have end-stage renal disease at the time of diagnosis ARCUS mediated knockout of HAO1 gene in liver Prevent buildup of oxalate Aim to develop a one-time, permanent treatment Combined liver-kidney transplant often required Rare genetic disease characterized by accumulation of calcium oxalate in kidneys, which leads to painful kidney stones and ultimately end-stage renal disease Our Approach Primary Hyperoxaluria – key facts

Effective, Efficient ARCUS Editing Underpins Advancement of PH1 ARCUS efficiently knocked-out the HAO1 gene in non-human primates following AAV8 delivery NHP 1 NHP 2 NHP 3 NHP 4 Day 128 Knock-out % ARCUS treatment resulted in ~70% reduction in urine oxalate in a PH1 mouse model 0 7 14 21 28 35 42 49 56 63 0 50 100 150 200 Day Urine oxalate (% baseline) High dose Low dose Buffer only EG* added to diet Mouse model Non-human primate (whole liver) *Ethylene glycol Data on file Dose: 3x1013 vg/kg

Feed the Planet Elo Life Systems

An Efficient Business Model A Human Health Opportunity Food companies need new inputs to respond to: Climate change Consumer preference Elo integrates ARCUS with enabling technologies to create greatly needed improvements to sources of food Partner driven Minimal capital investment Elo is Focused on Delivering Healthy and Sustainable Food Sources ELO LIFE SYSTEMS Discovery Proof of Concept Lead Generation PARTNER Field Testing Pre-Launch Product Launch

Multiple Upcoming Milestones Expected Across Portfolio in 2020 IND accepted for BCMA CAR T Initiate dosing for CD20 CAR T Initiate dosing for BCMA CAR T PH1 candidate selection NHL (CD19) clinical data update ALL (CD19) clinical data update ü

Key Takeaways Highly experienced team includes the pioneers in editing Proprietary ARCUS editing platform confers fundamental advantages Independent cGMP manufacturing capabilities Early allogeneic CAR T clinical data validate core strategy FY19 cash $181m, runway into 2H 2021; validating partnerships in each business area In vivo programs to address significant unmet medical needs

Dedicated To Improving Life
